What are DNA motifs and protein domains?
A common misconception is that DNA motifs and domains are referring to the same structures and patterns. A DNA motif is a short, recurring pattern in DNA that is assumed to have a biological function. These could be indicative of common functional binding domains, and therefore indicative of a similar function. Protein domains are sequences subject to pressures to maintain functional interactions. These may often result in specific interactions with DNA, other proteins, small molecules and metal ions.
Pyrin Domain Structures
|
|
Tripartite Binding Motif (TRIM) Family of Proteins
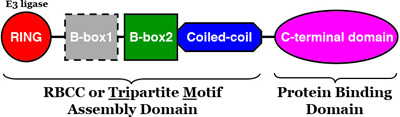
Pyrin protein product is part of a large family of proteins identified by their shared domain structure and functional capacity. Tripartite motif (TRIM) proteins share a common organization: an N-terminal RING domain, followed occasionally by a B-box 1, and then by a B-box 2 domain, a coiled-coil domain, and a C-terminal domain. The C terminal domain typically predicts where the protein product is binding, and is the most varied in TRIM proteins [1,2]. |
Figure 1. Tripartite motif (TRIM) protein domain structure [1].
|
|
TRIM proteins function through their RING domain, which typically function as E3 ligases and facilitate modification of target proteins with ubiquitin or ubiquitin-like proteins. Protein degradation is integral to normal function of the cell, and TRIM protein products are active from embryonic development into adulthood [1,2].
Pyrin Protein Structure |
|
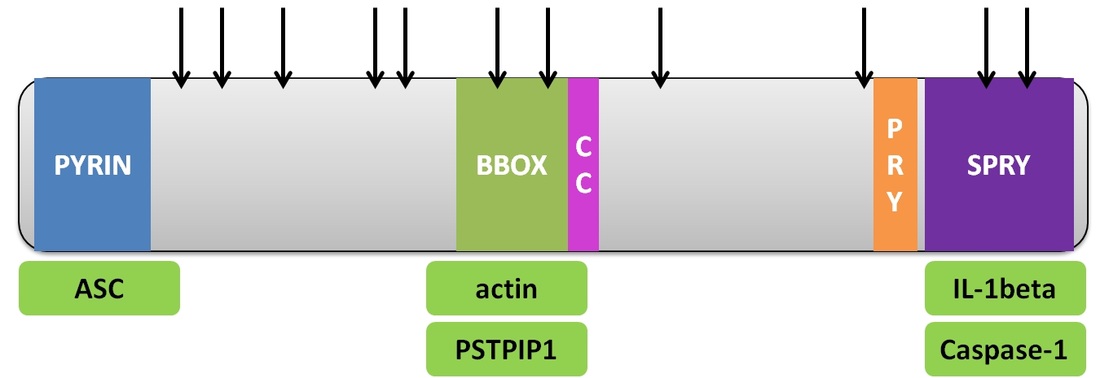
Like other TRIM proteins, pyrin has a tripartite structure that allows it to function within the cell. Pyrin protein has an N-terminal Pyrin domain (PYD), a member of the death effector-fold domains, a Zinc-finger B-box, a coiled-coil domain (CC), as well as a SPRY domain (also called B30.2 domain) [3].
Many of pyrin's domains have several known protein interactors shown in Figure 1. The PYD and SPRY domain interact with inflammasome components, and inhibit pro-IL1beta processing in the cell (see Protein Intro Page) [3]. The B-box domain interacts with actin at the leading edge and PSTPIP1, a protein which stabilizes actin [4]. |
Figure 2. Pyrin protein structure with domains Pyrin Domain (PYD), B-box 2, coiled coil, PRY and SPRY labeled, fibromyalgia associated missense mutations labeled with arrows above the protein [5], and known interactors with each domain listed below the domain.
|
|
Domain Identification
Protein domain identification tools can compare a protein sequence to a database of known domain architectures to identify matches. Some also analyze patterns, profiles and identify functionally critical amino acids to enhance their discriminatory power. In this analysis, SMART, Pfam, and PROSITE will be used to identify functional domains in the pyrin protein product. |
|
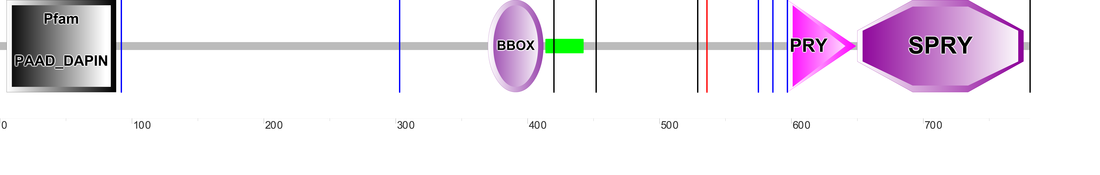
Figure 3 depicts the domain structures identified for pyrin in the SMART database and their amino acid locations along the protein. SMART was able to detect all five functional elements in the submitted FASTA sequence. Pfam was not able to identify the coiled coil region; and PROSITE only identified the DAPIN (PYRIN), ZF_BBOX, and B302_SPRY (580-775 aa). It appears that PROSITE was able to identify both C terminal domains, but lumped them together under the name B30.2.
|
|
Analysis
All three databases examined output similar domain results, though the comparison aided in annotating the many names of some domains, precise location, and how they function within the cell. Comparing the domains to those described in the literature was also a useful tool in determining domain function and interactions with other proteins. |
Pyrin Isoform 2
|
The MEFV gene can be alternatively spliced into a second protein product, the pyrin isoform 2. To analyze the functional differences between each protein, the FASTA sequence for this alternative isoform was also examined using SMART, Pfam and PROSITE.
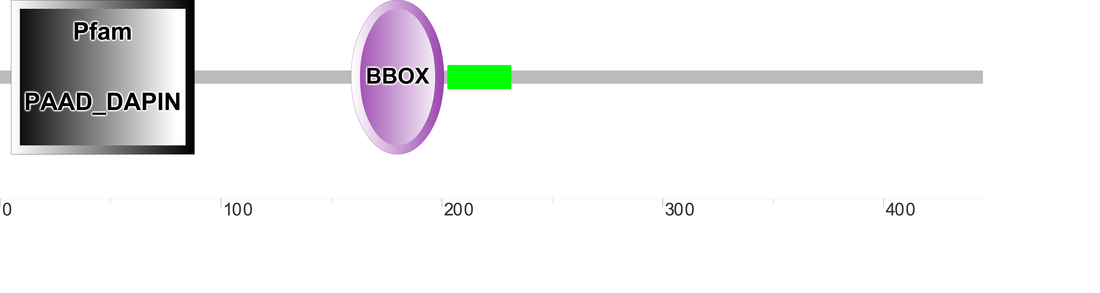
Pyrin isoform 2 is a much shorter protein product (445 amino acids), and contains Pyrin Domain (PYD), BBOX, and coiled coil regions. There are also fewer amino acids separating the N terminal PYD and the following BBOX domain than in the focal isoform of pyrin. This could signify the removal of an exon that does not code for a functional domain. |
|
Similar to the pyrin isoform 1, there were minor differences observed when comparing the results from the three databases. SMART was able to identify all three functional regions; and both Pfam and PROSITE only identified the PYRIN and BBOX domain. This is consistent with isoform 1, and may implicate isoform 2 in having a potentially different function in vivo.
|
References
[1] Pornillo, O. Pornillo's Lab. Last updated 29 Aug 2011. http://people.virginia.edu/~owp3a/research.html (accessed 7 Apr 2013).
[2] TRIM Family Gene. Last updated April 2011. http://ghr.nlm.nih.gov/geneFamily/trim (accessed 17 April 2013).
[3] Papin, S, Cuenin, S, Agostini, L, Martinon, F, Werner, S, Beer, HD, Grutter, C, Grutter, M, and Tschopp, J. (2007) The SPRY domain of Pyrin, mutated in familialMediterranean fever patients, interacts withinflammasome components and inhibits proIL-1b processing. Nature: Cell Death and Differentiation 14:1457-1466.
[4] Shoham, NG, Centola, M, Mansfield, E, Hull, KM, Wood, G, Wise, CA, and Kastner, DL. (2003). Pyrin binds the PSTPIP1/CD2BP1 protein, defining familial Mediterranean fever and PAPA syndrome as disorders in the same pathway. Proc Natl Acad Sci U S A. 100(23): 13501-13506.
[5] Feng J, Zhang Z, Li W, et al. (2009) Missense mutations in the MEFV gene are associated with fibromyalgia syndrome and correlate with elevated IL-1beta plasma levels. PLoS One 30: 8480.
[1] Pornillo, O. Pornillo's Lab. Last updated 29 Aug 2011. http://people.virginia.edu/~owp3a/research.html (accessed 7 Apr 2013).
[2] TRIM Family Gene. Last updated April 2011. http://ghr.nlm.nih.gov/geneFamily/trim (accessed 17 April 2013).
[3] Papin, S, Cuenin, S, Agostini, L, Martinon, F, Werner, S, Beer, HD, Grutter, C, Grutter, M, and Tschopp, J. (2007) The SPRY domain of Pyrin, mutated in familialMediterranean fever patients, interacts withinflammasome components and inhibits proIL-1b processing. Nature: Cell Death and Differentiation 14:1457-1466.
[4] Shoham, NG, Centola, M, Mansfield, E, Hull, KM, Wood, G, Wise, CA, and Kastner, DL. (2003). Pyrin binds the PSTPIP1/CD2BP1 protein, defining familial Mediterranean fever and PAPA syndrome as disorders in the same pathway. Proc Natl Acad Sci U S A. 100(23): 13501-13506.
[5] Feng J, Zhang Z, Li W, et al. (2009) Missense mutations in the MEFV gene are associated with fibromyalgia syndrome and correlate with elevated IL-1beta plasma levels. PLoS One 30: 8480.