Pyrin
The human MEFV gene is composed of 10 exons and encodes a 781 a.a. cytoplasmic protein typically called pyrin. Pyrin protein has five functional domains: DAPIN/pyrin, b-Zip, B-Box, coiled-coiled, and PRY/SPRY(B30.2) (further described in Protein Domain).
In humans, there are two isoforms of pyrin; this analysis focuses solely on isoform 1. Additionally, it is important to note that pyrin is known by many names including FMF, MEF, MGC126560, MGC126586, TRIM20, and marenostrin; this website will only use pyrin for consistency. Pyrin and Inflammation |
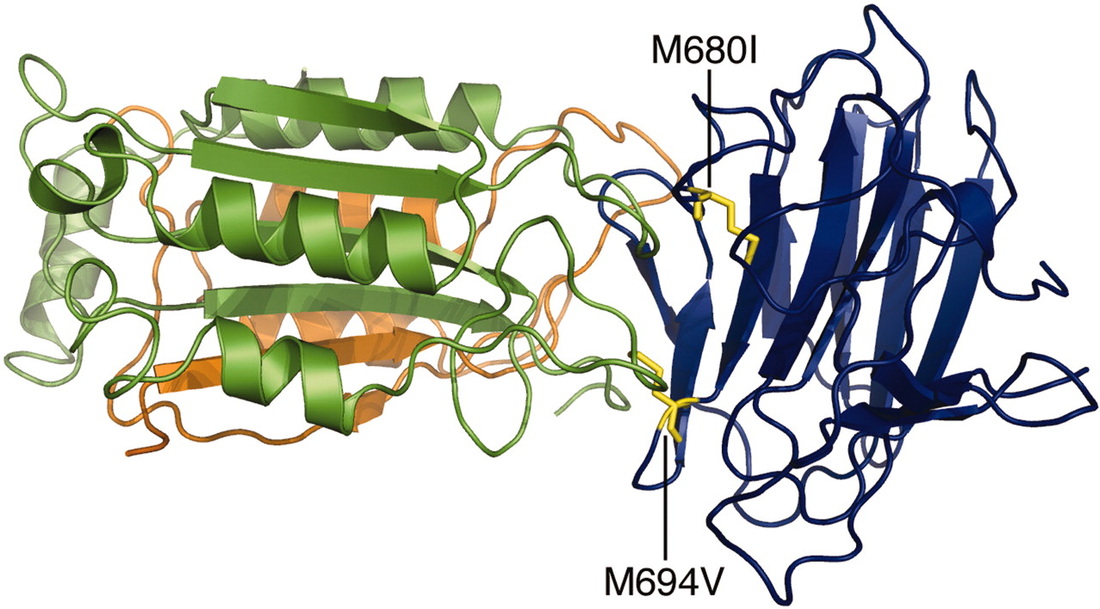
Figure 1. An image of the B30.2 binding domain in pyrin (Chae et al. 2006)
|
|
Inflammation of tissue happens when the immune system response sends white blood cells (WBC) and signaling molecules to a site of injury or disease. These messengers will begin to fight foreign invaders and facilitate tissue repair if there has been damage. After the body has accomplished these goals, the inflammatory response must be "turned off" to prevent damage to host tissue and cells. In humans, pyrin is produced in certain WBCs that play a role in inflammation and fighting infection, like neutrophils and monocytes [1]. The exact mechanism of pyrin is not known, but it has been shown to play a key role in the "inflammasome" and in its interactions with the cellular cytoskeleton.
|
|
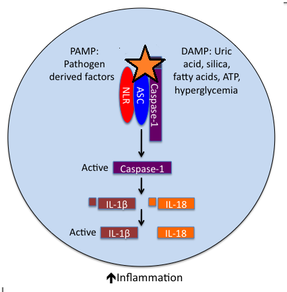
The "inflammasome" are a compilation of proteins which initiate the inflammatory response by their assembly and oligomerization into a common structure. Triggered by signaling processes, these protein complexes mediate the caspase-1-dependent processing of cytokines. This cascade leads to the maturation and release of pro-inflammatory cytokines, especially IL-1β and IL-18, and the initiation of the inflammatory response [2,3]. The SPRY domain of pyrin was shown to be a regulator for inflammatory response, interacting with not only inflammasome proteins, but caspase-1 and its substrate, proIL-1β [4]. Evidence showed that though pyrin knock-down increased inflammatory response, overexpression of SPRY domain alone blocks those processes [4]. Additionally, pyrin binds to another inflammasome protein, ASC, by means of its PYD domain. The mechanism of association and result on caspase-1 activity has remained controversial, likely due to the different model systems in use [4].
To further characterize pyrin's role in the cell, it has been demonstrated that pyrin protein is localized to the leading edge in monocytes, where is co-localizes with polymerizing actin. Similarly, pyrin becomes localized when inserted into bacteria with dynamically polymerizing actin-rich tails. The last functional domains, the B-box and coiled-coil regions, are required for association, and though it does not appear to have an affect on disease causing mutations, it could have unknown functional consequences on migration and cell synapse formation [5].
|
Figure 2. The Inflammasome Cascade. By binding ASC and other inflammasome proteins, caspase-1 is activated to cleave proIL-1β and proIL-18. The orange star shows the interaction of pyrin in this system, binding to ASC and associating with caspase-1 and pro-cytokeins.
|
Evolution of a protein
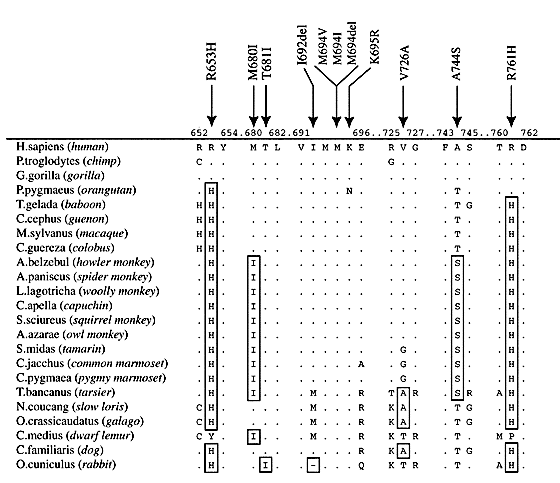
Figure 3. Partial protein sequence alignment for 20 primates and 2 non-primate mammals. Human amino acid (aa) sequence is listed at the top. Dots infer the same aa, whereas changes are marked by their new aa in the sequence. Arrows indicate the aa which are described as locations of missense mutations in Mediterranean Fever patients. Figure from Schaner et al. (2001).
|
Learning vocabulary
A protein can be influenced by evolutionary processes in both the positive and negative direction. Positive selection favors rare alleles that infer an evolutionary advantage, whereas negative selection would favor a predominant allele to stabilize in the population. By examining amino acid changes in sequence homology, one can potentially infer the direction of selection. Pyrin Evolution in Humans In the population, the rare alleles of pyrin harbor missense mutations and are known to be associated with diseases like fibromyalgia and Familial Mediterranean Fever. Knowing this, one would expect these alleles to be very uncommon in the population. However, when examining Middle Eastern populations, we see carrier frequencies as high as 1:3 [6]. This may be due to a heterozygote advantage for mutant pyrin carriers. In studies comparing human pyrin sequence homology with other primates, mutations in the SPRY binding domain in pyrin has been shown to be under positive selection (Figure 3) [6]. This further supports the potential heterozygote advantage of pyrin. In all disease models to date, missense mutations have been responsible for the inhibited function of mutant protein products. No null, or complete lack of function, products have been found. This suggests that pyrin function is important for survival of the organism. |
References
[1] MEFV. 2008. http://ghr.nlm.nih.gov/gene/MEFV
[2] Schroder and Tschopp. (2010). The Inflammasomes. Cell 140 (6): 821–832.
[3] Wikipedia: Inflammasome. http://en.wikipedia.org/wiki/Inflammasome (accessed 2 Feb, 2013).
[4] Papin, Cuenin, Agostini, Martinon, Werner, Beer, Grütter, Grütter and Tschopp. (2007). The SPRY domain of Pyrin, mutated in familial Mediterranean fever patients, interacts with inflammasome components and inhibits proIL-1 processing. Cell Death and Differentiation 14: 1457-1466.
[5] Waite, Schane, Hu, Richards, Balci-Peynircioglu, Hong, Fox and Gumucio. (2009). Pyrin and ASC Co-Localize to Cellular Sites that Are Rich in Polymerizing Actin. Experimental Biology and Medicine 234 (1): 40-52.
[6] Schaner, Richards, Wadhwa, Aksentijevich, Kastner, Tucker, and Gumucio. (2001). Episodic evolution of pyrin in primates: human mutations recapitulate ancestral amino acid states. Nature Genetics 27: 318-321.
[1] MEFV. 2008. http://ghr.nlm.nih.gov/gene/MEFV
[2] Schroder and Tschopp. (2010). The Inflammasomes. Cell 140 (6): 821–832.
[3] Wikipedia: Inflammasome. http://en.wikipedia.org/wiki/Inflammasome (accessed 2 Feb, 2013).
[4] Papin, Cuenin, Agostini, Martinon, Werner, Beer, Grütter, Grütter and Tschopp. (2007). The SPRY domain of Pyrin, mutated in familial Mediterranean fever patients, interacts with inflammasome components and inhibits proIL-1 processing. Cell Death and Differentiation 14: 1457-1466.
[5] Waite, Schane, Hu, Richards, Balci-Peynircioglu, Hong, Fox and Gumucio. (2009). Pyrin and ASC Co-Localize to Cellular Sites that Are Rich in Polymerizing Actin. Experimental Biology and Medicine 234 (1): 40-52.
[6] Schaner, Richards, Wadhwa, Aksentijevich, Kastner, Tucker, and Gumucio. (2001). Episodic evolution of pyrin in primates: human mutations recapitulate ancestral amino acid states. Nature Genetics 27: 318-321.